Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous in the environment, with one of the major sources being the petroleum industry. PAHs are often concentrated in urban environments because of increased urbanization and automobile use. They move through the environment via stormwater runoff, atmospheric transport, municipal and industrial wastewater, and combustion of organic material (Meador 2008). PAHs are divided into low and high molecular weight compounds based on the number of benzene rings in the molecule. There is also a distinction between parent compounds with a basic ring structure, and alkylated homologs, which can be as toxic or more toxic than the parent compound (Meador 2008).
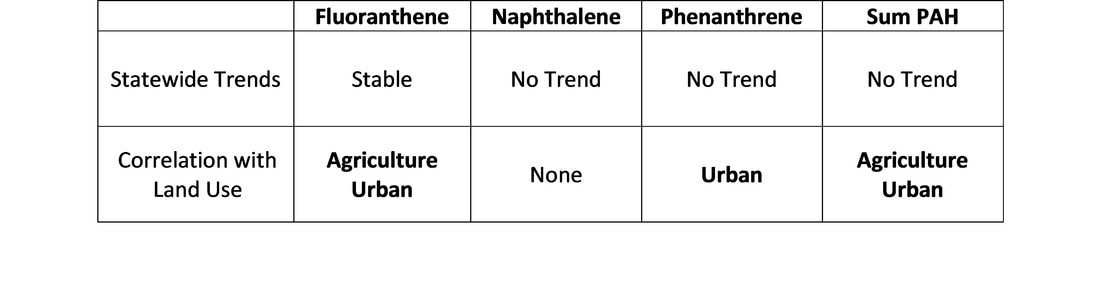
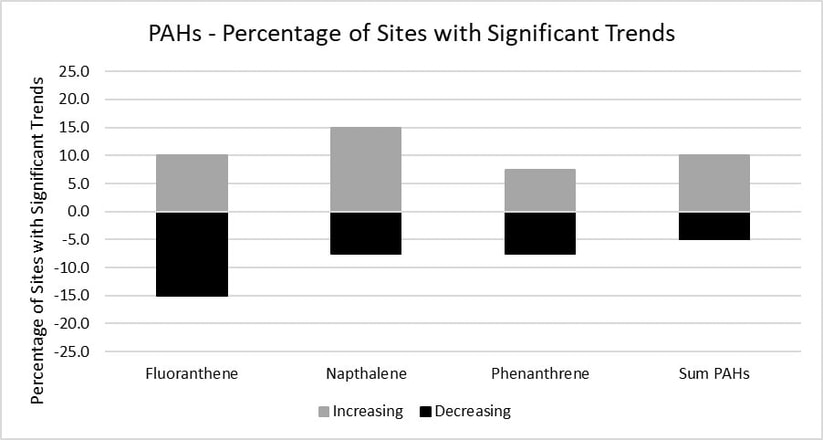
Trends were calculated for the Sum of PAHs, as well as the individual compounds fluoranthene, naphthalene and phenanthrene at both a statewide scale and at individual sites. Trends were calculated from 2010 to 2021 because there are significantly lower sample sizes for 2008 and 2009 for current urban sites. The sum calculation was made up of the following compounds: acenapthene, acenapthylene, anthracene, benz(a)anthracene, benzo(a)pyrene, benzo(b)fluoranthene, benzo(g,h,i)perylene, benzo(k)fluoranthene, dibenz(a,h)anthracene, chrysene, fluoranthene, fluorene, indeno(1,2,3-c,d)pyrene, naphthalene, phenanthrene, pyrene. Data from 2018 to 2020 were omitted because of an anomalous number of non-detects (45.3% of samples without a single detected PAH compound), likely caused by errors in chemical analyses.
Statewide trends for three individual PAH compounds and the Sum PAH were stable or did not show significant increases or decreases. A similar number of individual sites showed significant increases and decreases with no clear trend based on individual compounds or the Sum PAH.
Reference
Trends were calculated for the Sum of PAHs, as well as the individual compounds fluoranthene, naphthalene and phenanthrene at both a statewide scale and at individual sites. Trends were calculated from 2010 to 2021 because there are significantly lower sample sizes for 2008 and 2009 for current urban sites. The sum calculation was made up of the following compounds: acenapthene, acenapthylene, anthracene, benz(a)anthracene, benzo(a)pyrene, benzo(b)fluoranthene, benzo(g,h,i)perylene, benzo(k)fluoranthene, dibenz(a,h)anthracene, chrysene, fluoranthene, fluorene, indeno(1,2,3-c,d)pyrene, naphthalene, phenanthrene, pyrene. Data from 2018 to 2020 were omitted because of an anomalous number of non-detects (45.3% of samples without a single detected PAH compound), likely caused by errors in chemical analyses.
Statewide trends for three individual PAH compounds and the Sum PAH were stable or did not show significant increases or decreases. A similar number of individual sites showed significant increases and decreases with no clear trend based on individual compounds or the Sum PAH.
Reference
- Meador JP. 2008. Polycyclic Aromatic Hydrocarbons. In: Jørgensen SE, Fath BD, editors. Encycolpedia of Ecology. Amsterdam, Netherlands: Elsevier Science. p. 3120.